
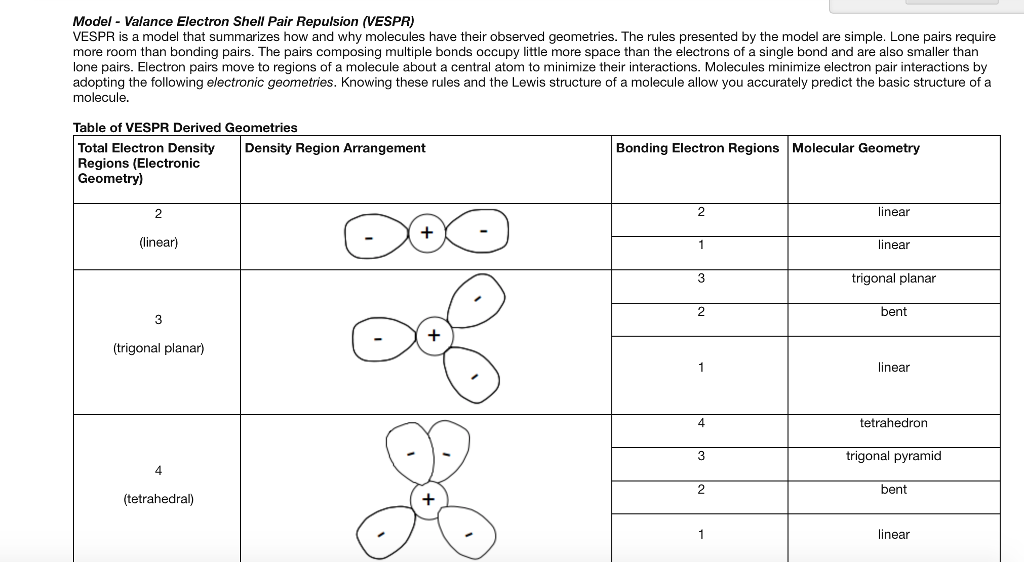
This arrangement determines the geometry and shape of the molecule as well as the bond angle. VSEPR is based on the premises that there is repulsion between lone pair of electrons and always atom will arrange itself in that manner where there is minimum repulsion. VSEPR was first presented by Sidgwick & Powell in 1940. VSEPR stands for the Valance Shell Electron Pair Repulsion Theory. Prediction of geometry/shape (VSEPR Theory) Before VSEPR, lewis structure is used to predict the shape of the molecule. By changing the angle due to lone pair the geometry and shape of the molecule change.So, both exhibits opposite character and does not cancel out eachother effect so possess a dipole moment. For example in NH3: a single lone pair is present on the nitrogen atom which exhibits a negative character but hydrogen exhibits a positive character. A lone pair is also used to create a dipole moment.Further illustration occurs in VSEPR theory. The presence of lone pair decreases the bond angle because of higher electronic repulsion and higher electrical charge between the lone pair and the bonding pair of electrons. Therefore, the lone pair exhibits a polar character. A lone pair is involved in changing the angle of the molecule.The lone pair is involved in determining the geometry and shape of the molecule, and it is an essential part of the electron domain. Valance electrons in atom = Lone pair + Bond pair To calculate the number of valance electrons in an atom, we add lone pair in the bonding electron pair. We can identify the lone pair by using the Lewis structure. It is also called a non-bonded electron pair or unshared pair of electrons. It is connected to an atom by using a coordinate covalent bond. A pair of electrons that is not reacted or shared by an atom is called lone pair. lone pair and bond pair What is Lone pair?Ī lone pair is associated with the negative charge. The diagram represents the electron domains i.e. So, the repulsion of electrons is not only the factor that describes molecular geometries. electrons and positively charged nuclei attract towards eachother. 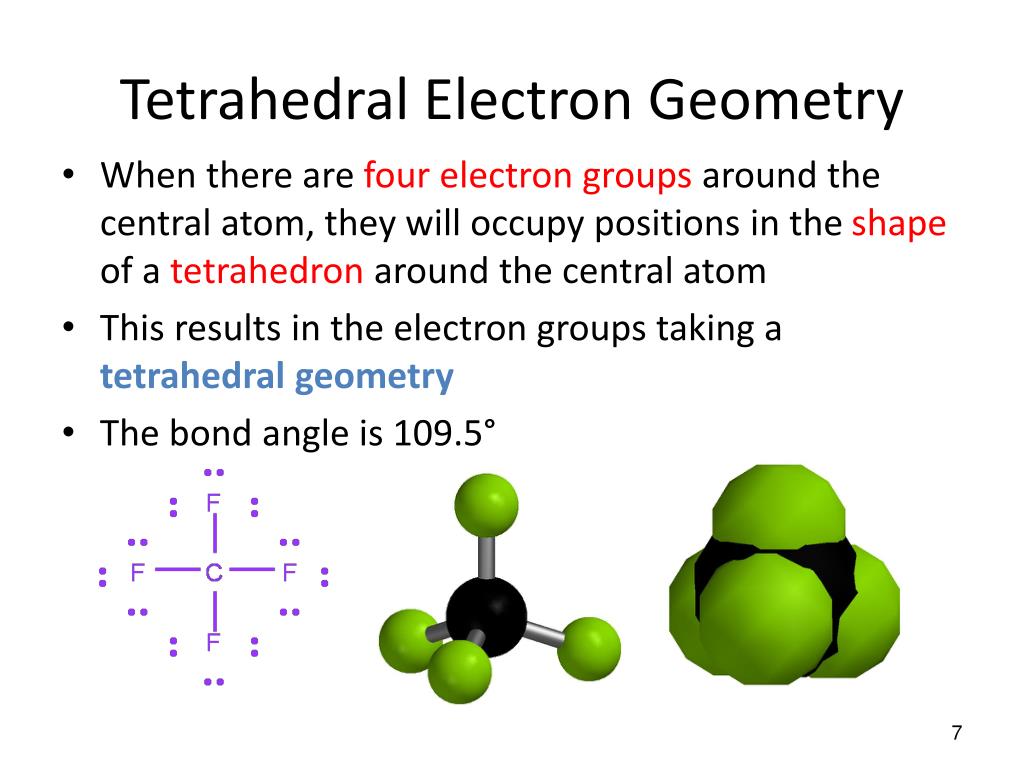
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
